Creating models of the intricate human brain is a daunting task, but tiny round structures assembled from brain cells demonstrate many qualities of the tissue within our skulls. Models developed in the Hoffman-Kim Lab even contain networks of miniscule vessels that resemble capillaries like those that carry blood through our brains. By constructing models that mimic the brain, scientists can better understand neurological problems and develop treatments for them more effectively.
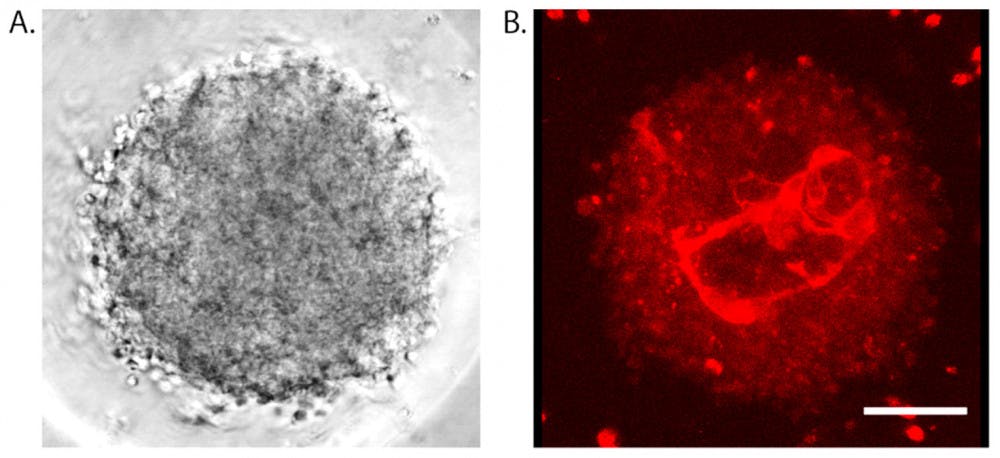
These mini model brains were born in the Hoffman-Kim Lab: Researchers placed cells from the brains of young rats in plates where they sank and then started sticking together, eventually forming a round mass of connected cells. “It typically takes about 24 hours for the cells to form a compact spheroid, and then they’ll grow from there,” said Molly Boutin, lead author of the study and a postdoctoral fellow at the National Center for Advancing Translational Sciences.
Despite their tiny scale, these models possess many characteristics of the human brain itself. New chemical treatments rendered the models more transparent so imaging could reveal the capillary-like networks within them, Boutin said.
The presence of these networks means that the models may help researchers study neurological issues. Many neurodegenerative diseases affect the blood vessels in the brain, said Liana Kramer ’17, co-author of the paper. “It’s really important to understand what happens in all these different disease states in order to make therapeutics and treatments.”
Recovery from a stroke or head trauma is linked to the growth of blood vessels in the brain, Boutin said. Observing blood vessel development in these models after injury might also yield insight for researchers.
Studying healthy models could be productive as well. “The brain is one of the body systems that isn’t well understood yet,” Kramer said.
Compared to many commonly used models, these structures have more in common with actual human brains. Instead of using just neurons, they include multiple cell types. Boutin said that the way these cells interact still eludes scientists, but studying them together will better illustrate what happens in the brain.
Scientists could feasibly use these models to test new therapies for neurological diseases. About 90 percent of drugs fail clinical trials, according to a study published in the journal Nature. “There’s obviously something wrong with that,” Boutin said. The cell-based models used to test drugs are one piece of that process. Models that do not reflect the operations of the body might show drugs failing when they could help real patients.
No drug for Alzheimer’s disease has passed clinical trials, for example. There are “many arguments for why they failed, but one is that human brains are different from animal brains,” said Hansang Cho, assistant professor of mechanical engineering and engineering science at The University of North Carolina. If scientists could build accurate models of human brains, they could provide more accurate drug testing.
Two-dimensional brain models — cells plated on glass or plastic chips — are still commonly used in research and testing. But “that’s not what my body is like, so it makes sense that the cells don’t want to behave the way that they do in my body,” Boutin said.
“A lot of the neuro-research is pretty slow in its development, and a lot of that is because of the platforms that are used,” Kramer said. These brain models, which can be produced in great quantities, have the potential to accelerate research.
“Fortunately, there is a resurgence of interest in 3D models,” said Damir Janigro, scientific director of Flocel Inc., a company whose mission is to develop a model for drug testing that better reproduces the behavior of the blood-brain barrier.
The models produced by University researchers still cannot imitate the brain perfectly. Unlike a normal brain, no fluid flows through the mini models, which means they cannot demonstrate the distribution of materials. And they lack many components of the membrane separating blood from the brain, Janigro said.
Through collaboration with other researchers, the lab hopes to create a device that would allow fluid to flow through the networks and tissues. The same process they used here could be applied to human cells, Kramer said, which would create models more similar to human brains. “There’s a lot of different directions we can take this,” she added.